Presentation
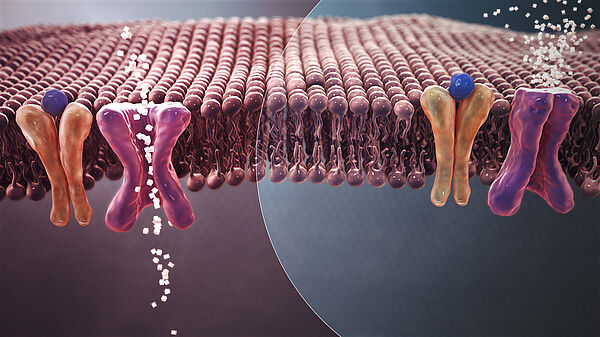
Because of ever increasing experimental data and knowledge, life scientists are now faced to highly complex biological systems with huge size. They involve many interactions that are more and more measurable both in time and space with possibly observable stochastic effects. Modeling and simulation are thus essential for the rational understanding of the dynamic behavior of living organisms. Besides of understanding, models are particularly valuable for their predictive power that allows for in-silico experiments. These aim at deciphering between costly and long wet lab experiments to be carried out.
The BioComputing group develops and studies formal methods and languages for modeling and simulation of biological systems. Our approach is mainly based on methods used for the static analysis of programs (within the fields of semantics of programming languages, logic and concurrency theory). A model is designed as a set of abstract interaction rules between biological entities possibly equiped with kinetic laws that specifies how long and how likely an interaction can occur. Such models, known as reaction networks, give rise to several interpretations of concurrency and time that may be non deterministic, deterministic (ODE systems), or stochastic (Markov chains). The choice of interpretation usually depends on the available knowledge and the biological question of interest.
Currently, our group is especially interested in developing methods that can give insights on biological behavior when only partial knowledge is available, in particular when the structure of the network is given but not (or few) kinetic parameters. Another line of research is the data acquisition from time lapse cell images based on machine learning techniques. These constitute the 3 projects described below.
BioComputing is a team of CRIStAL lab and member of the MSV thematic group.